Audit ready and Compliant Production
Audit ready and
Compliant Production
Audit ready and Compliant Production
Audit ready and
Compliant Production
Pharma industry is the most regulated industry in the world! Digital transformation of the Pharma sector is expected to reduce the cost, time and complexity of managing regulatory compliance. Paperless production, digital execution with digital BMR, predictive maintenance, IoT enabled smart sensors are all playing a vital role to help Pharma move into efficient and effective operations.
Change is a complex and expensive process in regulated environments. Digital transformation requires a very methodical approach with an execution strategy taking into account the inherent needs of the regulations. Pharma 4.0 can bring in great benefits to the industry but has to overcome the hurdles arising out of change management. Vegam’s experience and solutions help pharmaceutical companies address these key business challenges by providing a modular and scalable approach for digital transformation.
Read more
Audits by regulators No More a Pain
Digital Twin
Digital Twins are the future of manufacturing optimization and visualization. Demand for remote management of manufacturing has also fuelled the need for Digital Twins. Ability to simulate scenarios and visualize the implications is helping management aid in capital investments and strategic choices.
Vegam offers vDigiTwin platform wherein all the data related to production are unified, synced and structured. Relevant relations between data sources and systems is established so that the end-2-end flow of materials, operations and information is available with all the inter-relations & dependencies captured. Open APIs enable third party applications develop applications with access to structured data. Easy to develop real-time and offline visualization views using a drag & drop interface supported by vDigiTwin.
Schedule a Demo
Analytical Decision Making
Schedule a Demo
Digital transformation of manufacturing has resulted in generation of “information repositories” with data from various systems. Analysis of the information and associated facts can help un-earth insights that can predict future events. Artificial intelligence & Machine Learning techniques can be applied to develop predictive models which can generate prescriptive alerts & insights where in operations can be “Proactive” than “Reactive” to events.
Vegam Team brings expertise, skills and resources needed to work with the “Information repositories” and develop solutions that can generate data driven predictive triggers.
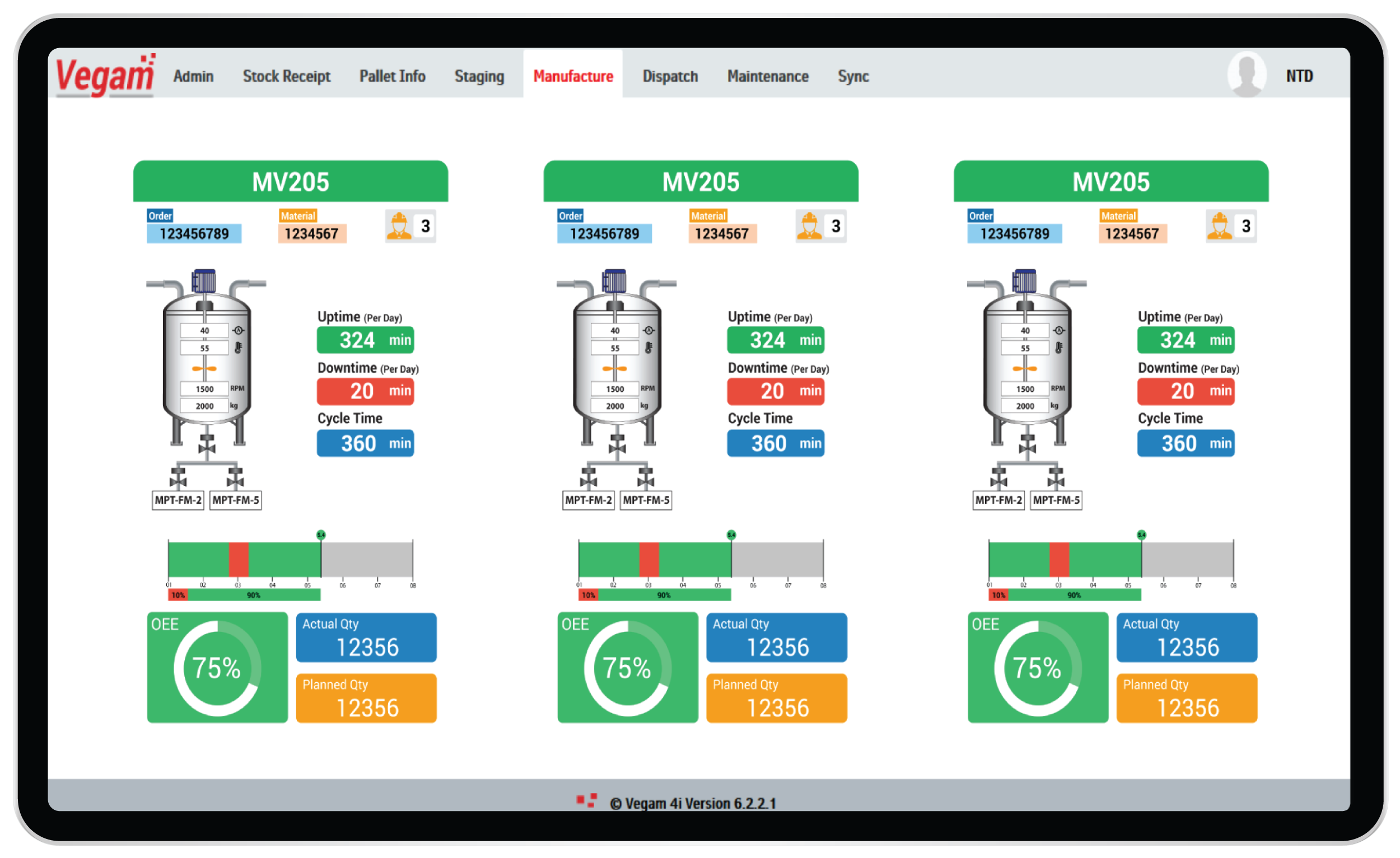
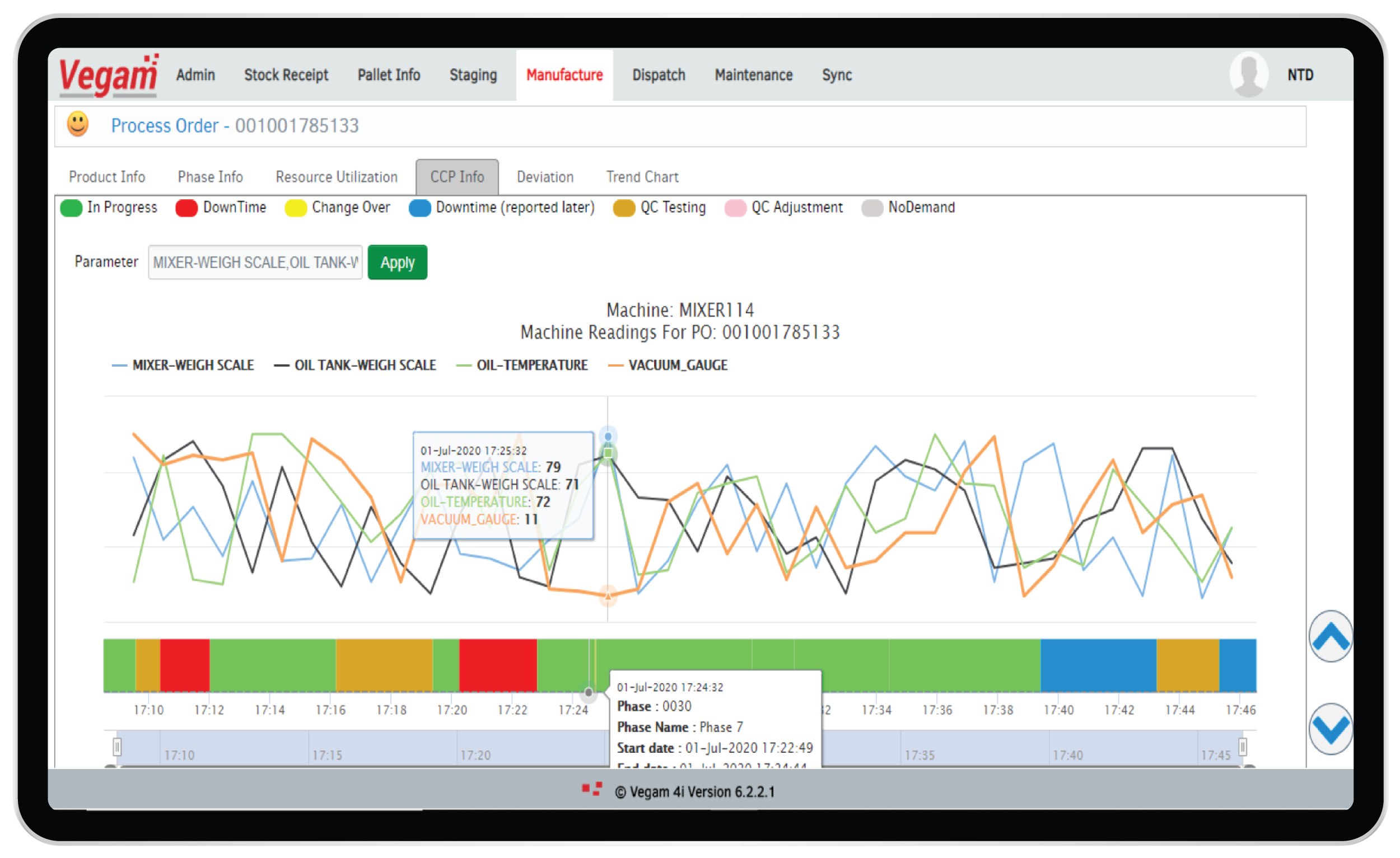
Digital Execution and BMR
Multiple applications and systems used by plants with extensive paper trails hamper the ability to have 360 degree, real time view of production operations & processes. This will fuel non-adherence to standard operating procedures with severe regulatory penalties. Proactive and reactive remedies are very difficult as the scale of operation increases.
Vegam SFS provides Digital Execution and digital BMR solution addressing all the challenges faced by Pharma manufacturers. Modular architecture helps to offer the solution by unifying multiple applications and extending the solutions as and when needed. Vegam SFS allows for contextual drill down of data across operators, processes, equipment, inventory and quality results, covering multiple systems. Vegam SFS offers real-time alerts on deviation to SOPs or parameters, giving ability to plants take reactive measures and avoid failures or process deviations.
The integrated data with secure trace logs helps generate Digital BMRs and other regulatory filings automatically. There is no scope for tampering data and hence improves the overall regulatory compliance. The effort and complexity to support regulatory audits also reduces significantly.
Schedule a Demo
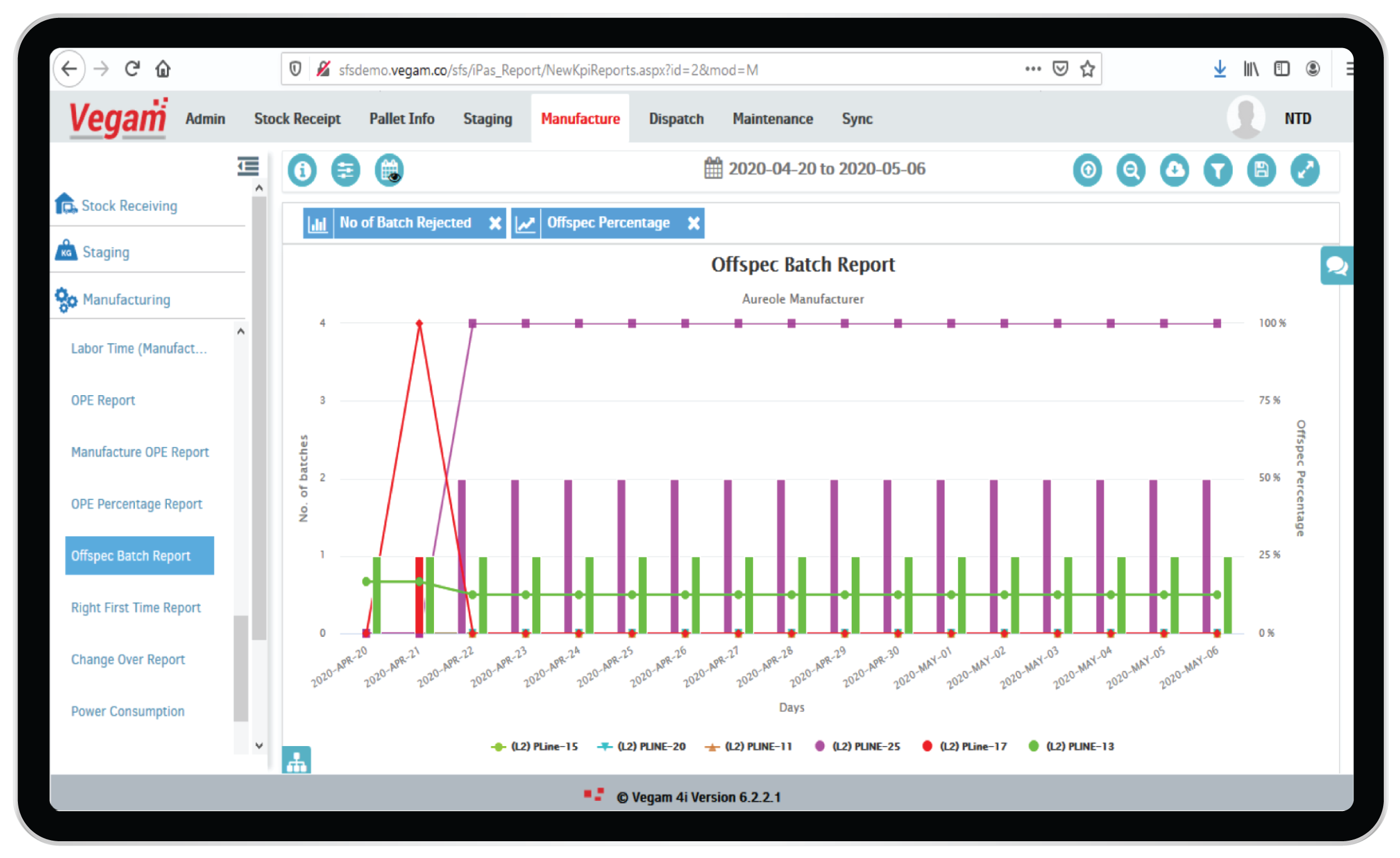
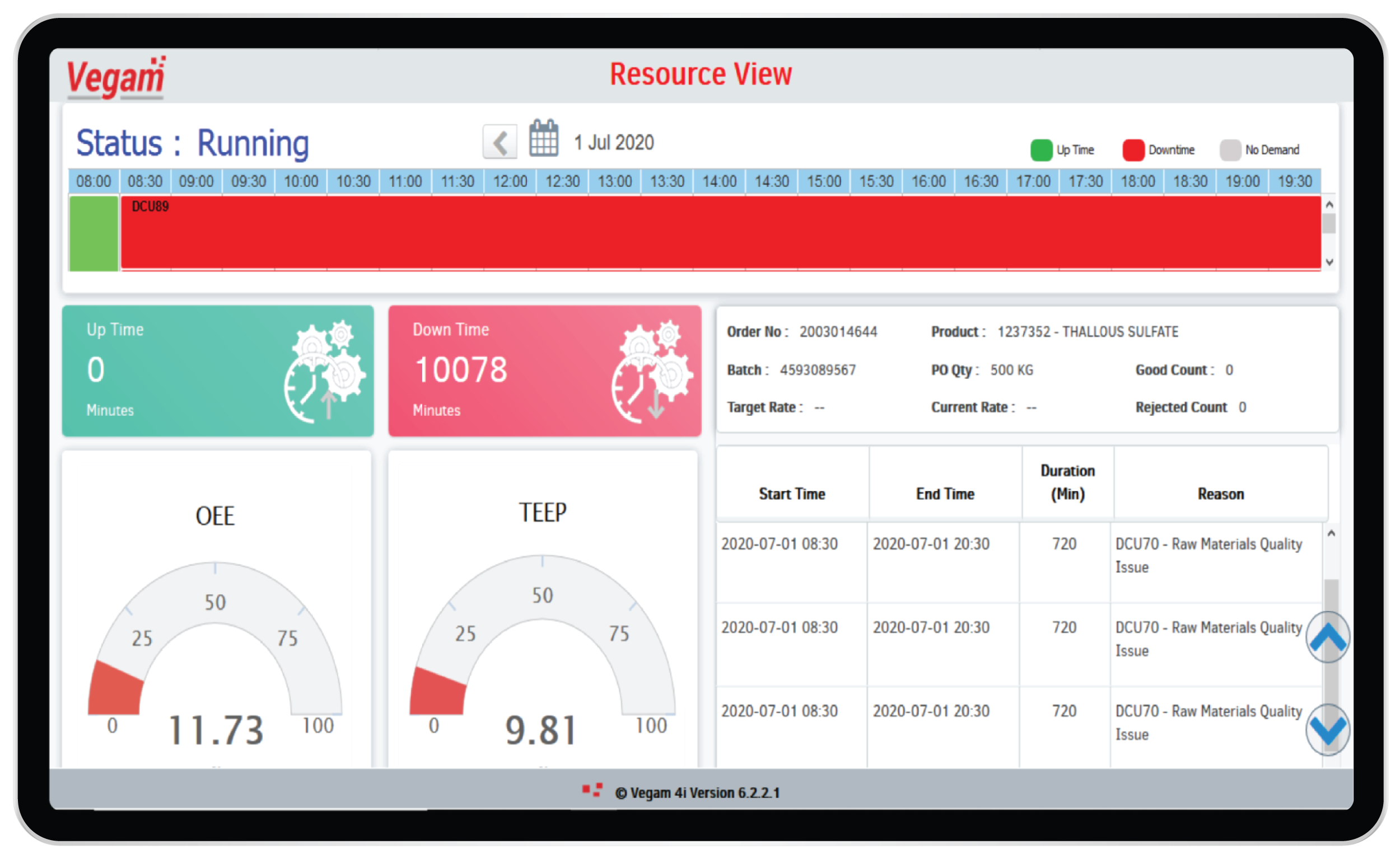
OEE & Productivity management
Schedule a Demo
Pharma manufacturers tend to offer multiple production lines with few shared resources among the production lines. Resources like centrifuge machines, mixers, bottling and filling units have different capacities and rates of operation. Human capital available on a daily basis would also define the rate at which production can happen in these plants.
Vegam SFS, SFS Lite and vPromax are different solutions and modules addressing the challenges to visualize in real-time the plant operations, performance and optimize the production. OEE calculations, down time measures, bottle neck resources analysis etc are tools to aid better planning and execution. Real-time KPIs and Dashboards with historical trends help in organizing effective daily morning meetings for better synergy among various operating teams. The solutions help in enhancing the overall health of the plant operations.
With the ability to seamlessly integrate across all technologies & systems of a complex pharma working environment, Vegam’s systems also help optimize production assets and their life and minimize spend.
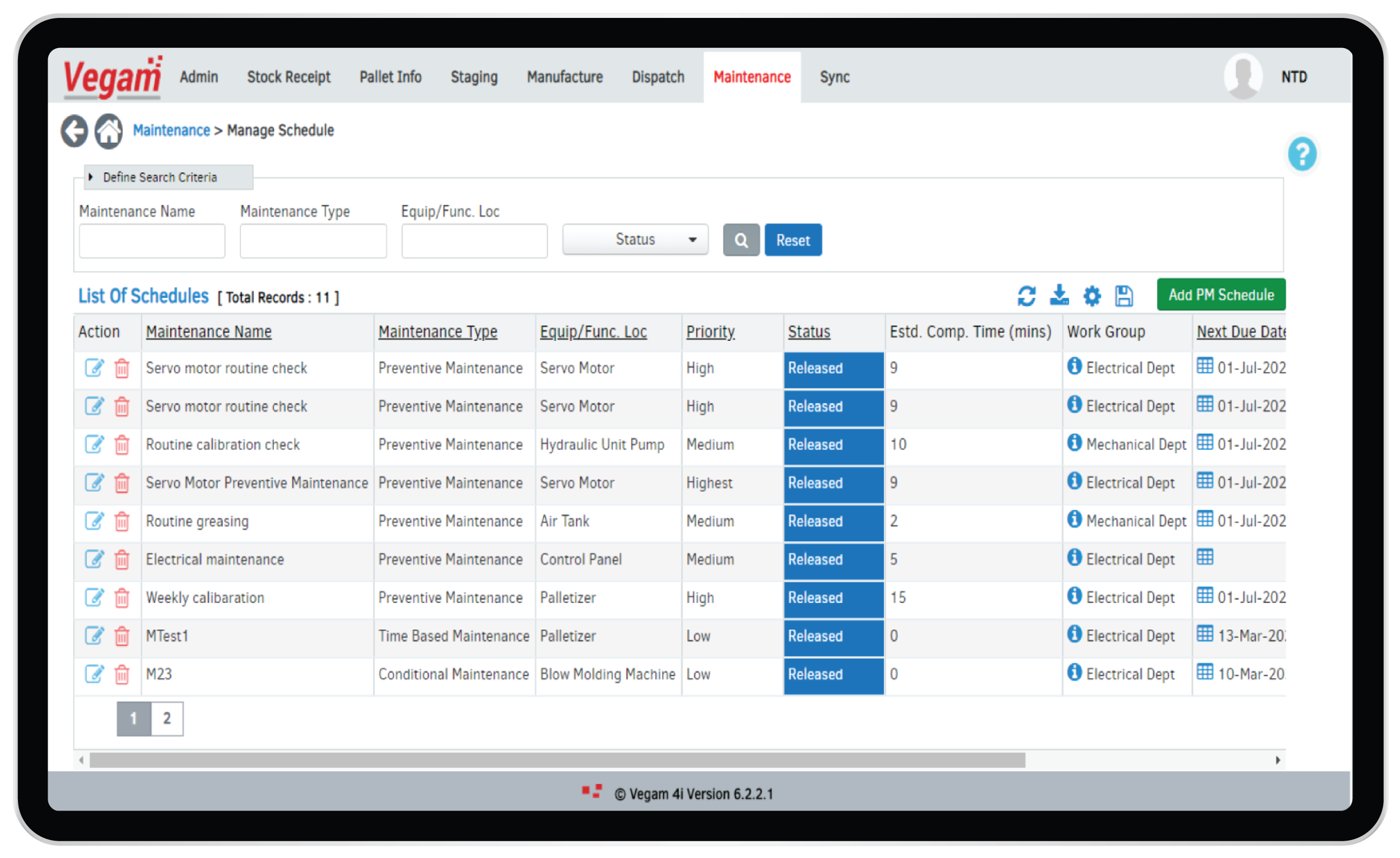
Maintenance Management
GMP expects strict SOPs followed with regards to asset maintenance and calibration. Preventive maintenance schedules, daily checklists and break down maintenance management are part of regulatory and compliance needs in Pharma production. Performing these task and recording the same is a large part of the operations at plants. Errors, discrepancies, delays can have serious implications to the business.
Vegam vMaint4.0, Vegam SFS & Vegam SFS Lite offer solutions and modules to support paperless management of all aspects involved in managing maintenance activities. Vegam addresses the core challenges in adopting offline or paper based solutions by bringing in mobility and real-time capabilities. Integrated with IoT, Vegam supports advanced capabilities like “Predictive maintenance”, Artificial Intelligence & Machine Learning based alerts and triggers to operate the plants in a proactive mode.
Benefits of paper-less, IoT based maintenance include:
- Improved team management and optimized workforce
- Adherence to Preventive Maintenance (PM) Schedules
- Spare Parts Management
- Real-time alerts and notifications
- Overall Equipment Efficiencies (OEE)
- Predictive Maintenance
- Supplier and Service provider intervention management.
Schedule a Demo
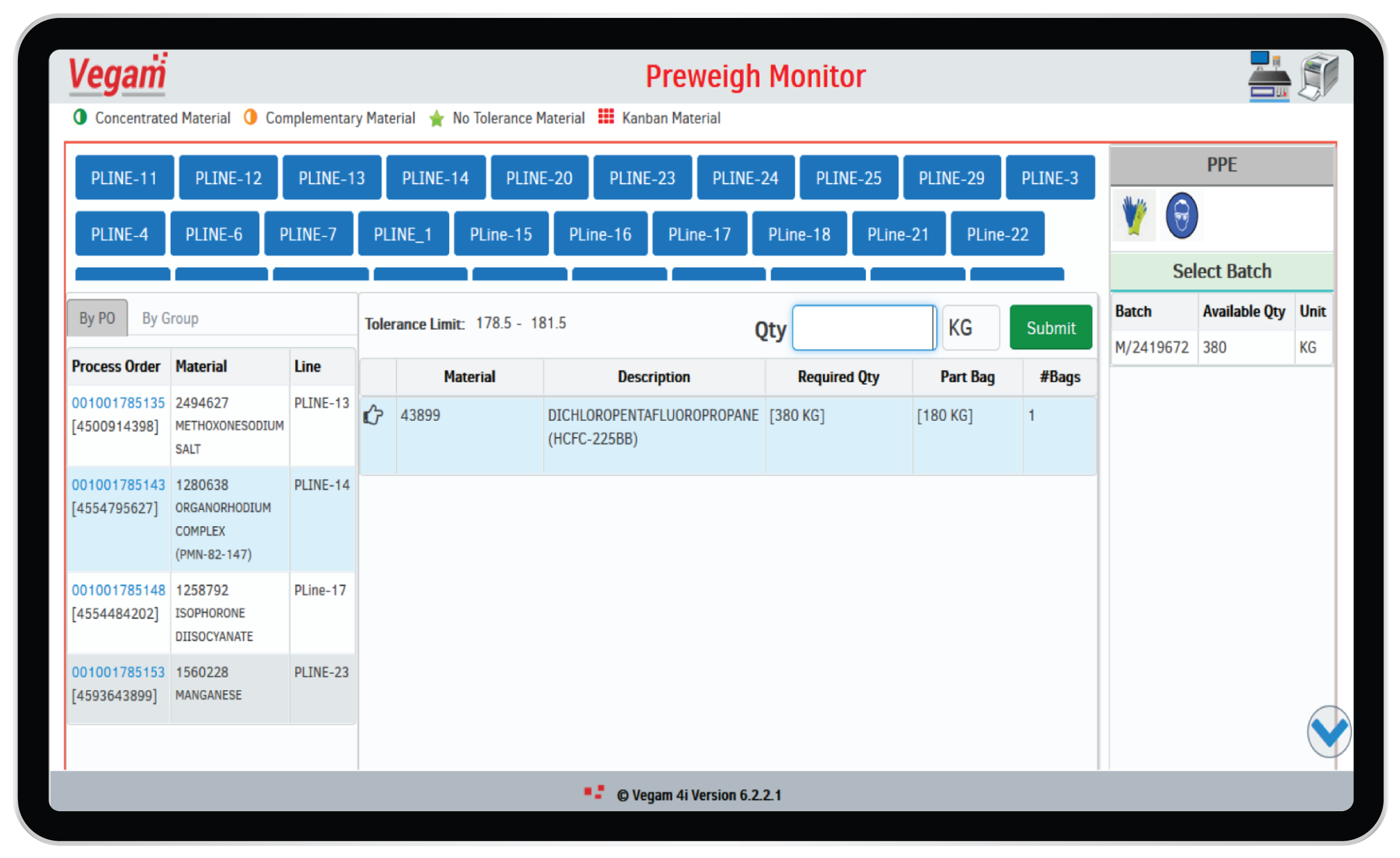
Material Management
Schedule a Demo
Pharma manufacturing has complex material management, starting from planning, procurement, storage, prestaging and staging to supporting right quality & quantity at the right place & time. This covers Receipt (including audits for very critical material) & quarantine, sampling & testing and storage of materials. Efficient material management can help:
- Drive efficient Production Scheduling
- Support material buying decisions & their quality control
- Receipt & inspection of material
- Supply & distribution of material
- Improve inter-departmental efficiencies
- Develop worker skills in material management
Current systems across small to large pharma manufacturing set ups are beset by paper trials and/or multiple complex systems. This makes it difficult to track and capture & display material data accurately and in real-time to support production and compliance (audit trails for very critical material) requirements. Apart from production halts, inability to track material data in warehouses results in heavy raw material and finished product losses.
Vegam Solutions’ Vegam SFS and vWarehouse helps pharma manufacturing plants track and capture material data accurately. This helps pharma companies comply to cGMP guidelines for Critical Material Management, have real-time inventory data ready for seamless production and reduce material losses.
One of the leading Adhesive
manufacturers in Asia was able
to achieve
10% Improvement
in productivity.
Customer success stories
Digital Transformation of Batch & Recipe based Manufacturing Industry
Case Study – Digital Transformation of Batch & Recipe based Manufacturing Industry Customer Profile Pioneer in Adhesive technologies...
Reach out to us
